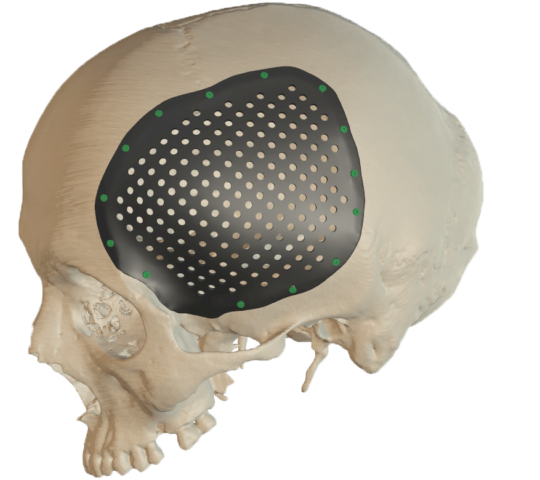
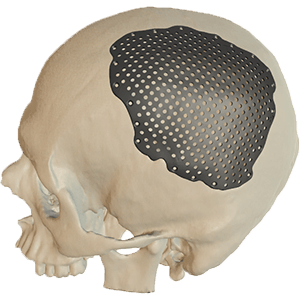
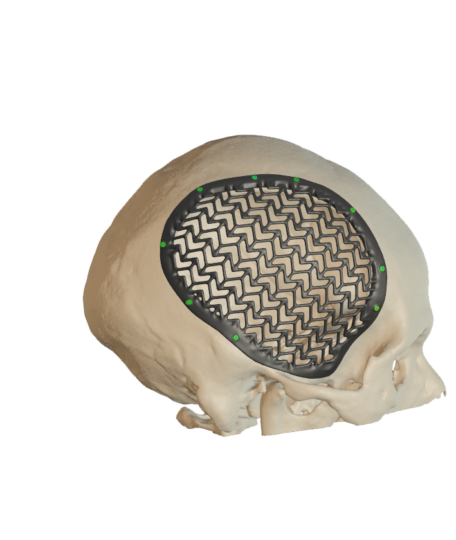
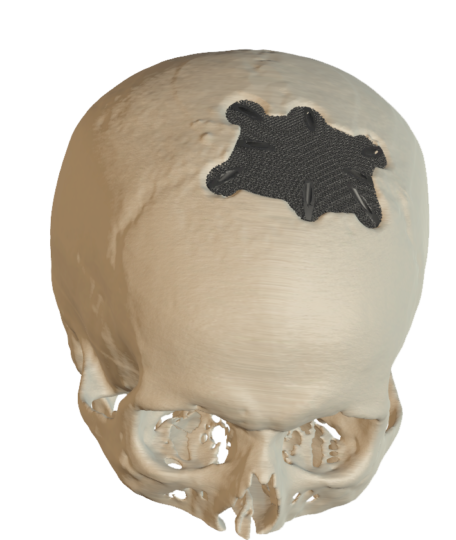
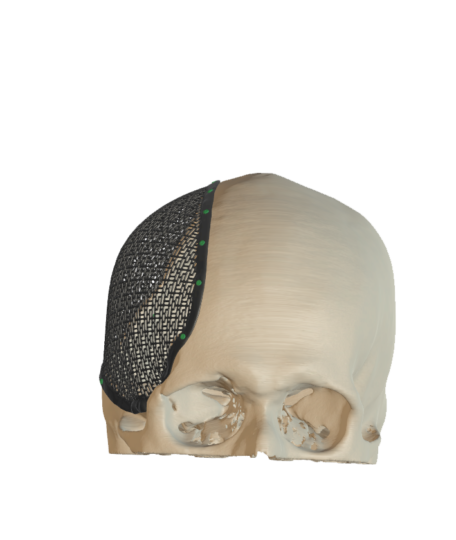
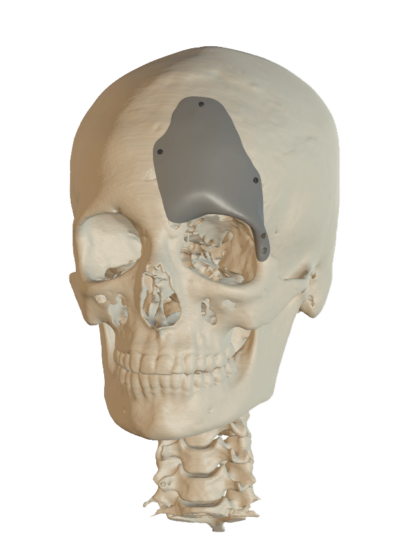
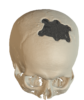
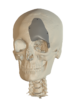
Cranial Plates
The anatomically shaped individual implant, available in a variety of biocompatible materials, guarantees excellent protection of intracranial contents and perfect restoration of the craniofacial contour.
This product is available in a variety of materials: Titanium mesh, solid titanium, PEEK
- Reduced surgery time
- Less risk of infection
- Anatomical reconstruction of the defect


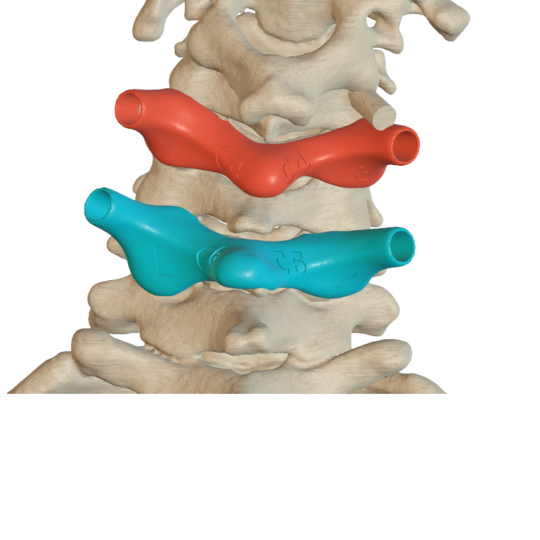
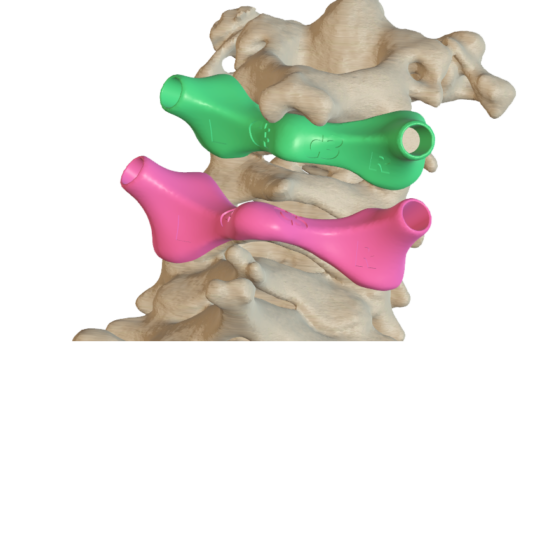
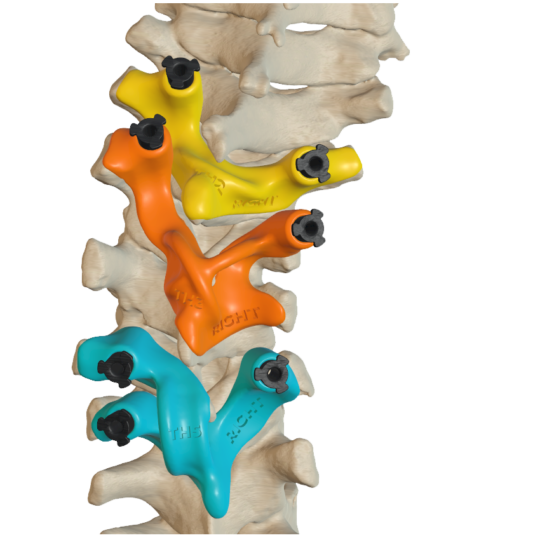
Surgical guides for vertebra
Individual guides for safe insertion of pedicle screws.
Preoperative planning and design by a qualified team ensures high safety and accuracy in use.
The guides are made of biocompatible material and contain metal inserts to keep the wound clean during surgical procedures.
- Reduced surgery time
- Less risk of infection
- Anatomical reconstruction of the defect






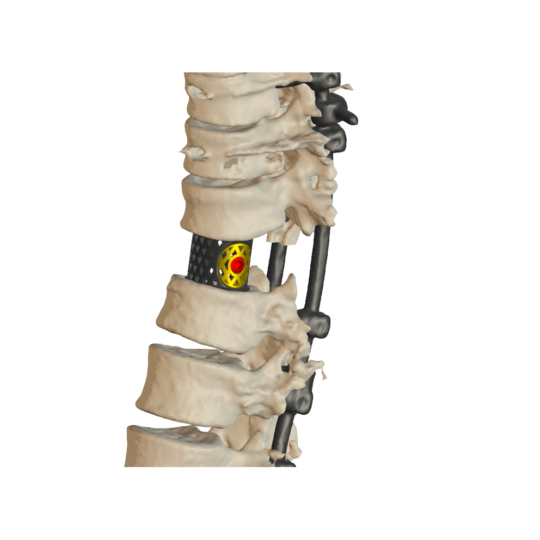
Individual spinal cages
sdadsadsadsad
- Reduced surgery time
- Less risk of infection
- Anatomical reconstruction of the defect



Materials

Ti6Al4V ELI
The best material for the creation of implants and other medical devices is currently considered to be titanium and its alloys, which have a full range of properties that meet the requirements of medicine: high corrosion resistance, sufficient mechanical and cyclic strength, and wear resistance.
Sterilization of products from titanium alloys by boiling, autoclaving, formalin vapor, burning, immersion in alcohol does not damage the metal surface.
Due to the biological inertness of titanium structures to the human body, during implantation, they are not rejected and do not provoke allergic reactions, quickly being covered with musculoskeletal tissues, the structure of which remains constant throughout subsequent life.
It can be used both in solid models and in the form of a titanium mesh, or a porous cellular structure for better osseointegration.
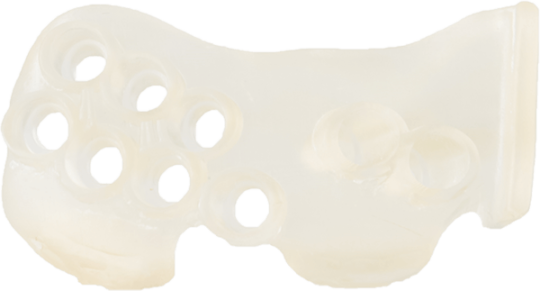
Photopolymer Resin
Biocompatible material, ISO 13485 compliant and designed for healthcare professionals worldwide. Such medical materials can be used for applications that require contact with the patient’s skin, mucosa and blood.
Used for the production of medical guides, fixation templates and sterilizable anatomical models

UHMWPE
UHMWPE has very low water absorption for organic polymer compounds, therefore the properties of UHMWPE do not change when exposed to water. The main properties of UHMWPE, which determine its use, are very high wear resistance, low coefficient of friction and high fracture toughness.
It is used for the production of titanium / polyethylene friction pairs

PEEK
PEEK is a synthetic material that has many advantages in CMF reconstruction surgery: strength, stiffness, chemical inertness and durability. PEEK is used for cranioplasty and facial implants.
The material can be sterilized and reused (in case of failure due to infection) and can be easily modified during surgery using surgical instruments.
PEEK, as a polymer, has an extremely low thermal conductivity compared to titanium, which already has a low thermal conductivity compared to other metals.